The selection of case studies shown below represents only a few examples of Mack’s medical manufacturing services and how they have helped customers resolve challenges and move product to market. It is not intended to provide a comprehensive listing of Mack’s involvement in any industry.
Stryker Case & Tray Delivery System
 As the knee system was being developed, Stryker also wanted to design an instrumentation set, including related cases and trays, that would accent the revolutionary implant. “We looked at a matrix of instrument users – surgeons, nurses, even cleaning and distribution staff – and configured cases & trays to satisfy their specific wish lists,” says Stryker Chief Engineer Scott Logan. “Then we talked to manufacturers whose bread and butter was making cases & trays. I was completely disillusioned with all of them because they were trying to force fit us into a generic package that made sense to them as manufacturers, but ignored the needs of our stakeholders.”
As the knee system was being developed, Stryker also wanted to design an instrumentation set, including related cases and trays, that would accent the revolutionary implant. “We looked at a matrix of instrument users – surgeons, nurses, even cleaning and distribution staff – and configured cases & trays to satisfy their specific wish lists,” says Stryker Chief Engineer Scott Logan. “Then we talked to manufacturers whose bread and butter was making cases & trays. I was completely disillusioned with all of them because they were trying to force fit us into a generic package that made sense to them as manufacturers, but ignored the needs of our stakeholders.”
At that same time, Stryker was already working with Mack Molding to co-develop ergonomic (coined Orthonomic™ by Stryker) overmolded handles for their top-of-the-line titanium instruments for the system. During a tour of its headquarters plant, Logan noticed Mack’s significant sheet metal fabricating capabilities in addition to its molding expertise “and realized that because we also do metal, we could produce a hybrid case & tray,” explains Larry Walck, Mack’s director of business development. “That was the beginning of a collaborative effort among Stryker designers and engineers and Mack process development and project managers that resulted in a distinctive delivery system to complete the knee implant package.”
The Challenge
Getting there wasn’t easy, however. The list of criteria was long. Based on Stryker’s 30-year orthopaedic history and input from surgeons and operating room (OR) staffs worldwide, “we were striving to incorporate everything our stakeholders needed into a modular case & tray set that was both lightweight and multi-tasking,” says Grecco.
Specifically, the delivery system needed to:
- Properly package the new flagship product by reflecting the same branding initiative, so that the Stryker system would be immediately recognizable by OR staff
- Consolidate the number of instruments and cases to use space more efficiently in the surgical theatre and minimize costly cleaning procedures
- Configure the instruments in stackable modules that correspond to surgical procedure and flow
- Withstand shipping and handling
- Remain under a specified weight for ease of handling
- Resist harsh chemicals used to meet worldwide cleaning standards
- Provide adequate steam flow during autoclaving to thoroughly clean instruments and eliminate wet-packing
- Provide instrument constraint with minimum contact for improved cleaning and autoclaving
- Stack in a sterility tub for shipping, which requires standardized sizes
- Avoid sharp edges that tear the sterile wraps and compromise the sterility of both the system and the surgical theatre.
“Time was another challenge,” adds Chris Wartinger, Mack program manager. “Cases & trays traditionally are the final piece of the puzzle. The design team has to finish the instruments and protocol first, which typically require some last minute tweaking to satisfy user needs. Those changes move down the line, impacting the delivery system as well. For example, if you make an instrument handle two millimeters larger, then we have to similarly adjust all brackets in all cases & trays, as well as all assembly prints. Consequently, we generally develop cases & trays when the rest of the program is 80 percent complete, leaving 100 percent of the work to do in 20 percent of the time.”
The Solution
The collaborative design team addressed the branding initiative with material, color and corporate identification. Titanium became the material of choice because it not only addresses several performance criteria, but also matches the instrumentation and distinguishes the sets from all others in the field. All plastic resin used on the instrument handles and case is custom formulated to match the Stryker brand. Both company and product identification are laser-cut into the case and molded into the instrument handles.
 The redesigned instruments feature a basic handle style with ergonomic soft-grip handles that are insert molded with a double overmold. The first material is a high heat polypropylene, and the second is a custom blended, thermoplastic elastomer (TPE).
The redesigned instruments feature a basic handle style with ergonomic soft-grip handles that are insert molded with a double overmold. The first material is a high heat polypropylene, and the second is a custom blended, thermoplastic elastomer (TPE).
To address the consolidation issue, the handle design introduces a quick attach-and-release mechanism that saves time in fixture assembly and minimizes the number of instruments required.
Downsizing the number of instruments allowed for consolidation of cases & trays as well. “In fact, we reduced the amount of cases required in the surgical theatre by 50 percent,” says Grecco, “saving space, an important commodity in the OR, and reducing costly cleaning procedures.”
The case & tray sets are two-tiered systems configured intuitively to correspond to the surgical procedure. They are laser-cut from titanium, a lightweight but durable material that withstands harsh cleaning solvents, as well as Stryker’s rigorous testing procedures.
Numerous aeration holes are laser-cut into the case to provide a sufficient flow of steam during autoclaving, as well as proper ventilation for drying. In addition, titanium brackets provide adequate instrument constraint with minimum contact for improved cleaning and autoclaving.
 “Mack uses laser welding and special forming techniques to support the instrumentation with titanium brackets,” adds Grecco. “We also anodize the titanium for an anti-galling effect and improved silk screening and cosmetics.”
“Mack uses laser welding and special forming techniques to support the instrumentation with titanium brackets,” adds Grecco. “We also anodize the titanium for an anti-galling effect and improved silk screening and cosmetics.”
The corners of the case are fitted with plastic bumpers molded of TPE for shock absorption and sterilization protection, since the TPE eliminates rough edges and corners. Plastic handles are also integrated into the trays. Again, both are color matched to the branding initiative, making the delivery system easily recognizable as a Stryker product, both in the OR and the marketplace.
Resources Required
 To better accommodate Stryker’s case & tray program, Mack invested $1.4 million to shorten the supply chain and lengthen its vertical integration in sheet metal fabrication, a core competency. “We added more laser-cutting capacity, as well as laser welding and other secondary operations,” says Walck. “The product is now manufactured almost completely in-house, which helps minimize risk in the supply chain. I can’t emphasize enough how important it is for OEMs to choose manufacturing partners who have the financial wherewithal and expertise to see them through the product launch, invest in their needs, and minimize all that can go wrong when dealing with multiple outside suppliers.”
To better accommodate Stryker’s case & tray program, Mack invested $1.4 million to shorten the supply chain and lengthen its vertical integration in sheet metal fabrication, a core competency. “We added more laser-cutting capacity, as well as laser welding and other secondary operations,” says Walck. “The product is now manufactured almost completely in-house, which helps minimize risk in the supply chain. I can’t emphasize enough how important it is for OEMs to choose manufacturing partners who have the financial wherewithal and expertise to see them through the product launch, invest in their needs, and minimize all that can go wrong when dealing with multiple outside suppliers.”
“Mack Molding is by far the most aggressive and enthusiastic organization I’ve every dealt with,” adds Grecco. “They made the capital investment to grow with us. The engineering resources were there. And they put in the time it took to get the job done. We built a relationship that’s growing in an exponential way.”
“We had very specific criteria for what we wanted,” concludes Logan, “and Mack met those criteria. That’s why I believe we’ve got a product that satisfies peoples’ needs. The success of this program really boils down to two words for me – collaborative effort.”
For more information, contact salesnorth@mack.com.
About Stryker Orthopaedics
Stryker Orthopaedics is a global leader in the development, manufacture and sale of orthopaedic products and services. Its hip, knee, upper extremity and trauma systems, as well as bone cement and bone substitutes, are designed to help patients lead healthier, more active lives. Stryker Orthopaedics is the largest subsidiary of the Stryker Corporation, which has delivered more than two decades of 20 percent annual net earnings growth.
Products referenced with ™ designation are trademarks of Stryker®.
……………………………………………………………………..
WaveMark Inventory Management System
The Power is in the Data
Optimized inventory. Expiration tracking. Accurate charge capture.
 “The power of WaveMark is not in the equipment — it’s in the data,” explains Carola Endicott, vp, hospital services, for the company she describes as an information business that helps people make better decisions.
“The power of WaveMark is not in the equipment — it’s in the data,” explains Carola Endicott, vp, hospital services, for the company she describes as an information business that helps people make better decisions.
Sure, WaveMark (Littleton, Mass.) is a self-described inventory management company that uses RFID technology to collect product usage information in real time. But it is equally, Endicott would argue, a point-of-use system and a business intelligence system. “The hospital manager who is on the hook to ensure that high-value medical products are managed well and efficiently is also on the hook to make sure she’s properly billing for the products used from each case,” says Endicott. “This is a really big problem for specialty procedural areas (cath labs, electrophysiology labs, interventional radiology labs, operating rooms) that rely on very busy clinical staff that aren’t necessarily trained in supply management to make sure inventory is properly documented.”
So how do WaveMark’s RFID cabinets help people make better decisions? “Yes, we’re good at counting,” says Endicott. “And we help hospital staff manage expired product and replenish product automatically. We save lots of time on the operational side. But the real dollar savings come from the data that allows staff to answer critical bottom-line questions:
- What IS the best deal I can get for this lab?
- Should I buy in bulk?
- Should I consign products?
- Should I reduce the variation of vendors?
At a time when cost reduction in health care could not be more critical, Endicott insists it is “definitely about the data.” Especially when 10-15 percent of the volume of products in a hospital represents 80-90% of the cost. Of the some 15,000 items on a hospital’s regular shopping list, about 85 percent cost $5-10 apiece. Syringes, drapes, bandages, products they buy a lot of. That leaves 2500 or so items that average around $800 each. So there’s a steep cost differentiation between the bundle of cheaper products and the expensive medical devices, where they need just enough, but not too much due to the cost burden.
How it works
That’s where WaveMark CIMS, a clinical inventory management system, comes in. RFID tags are placed on each item as it’s received through the hospital supply chain. Product information is captured from the product bar code, assigned to the RFID tag, and saved into the WaveMark CIMS database for real-time medical device tracking via the internet. No huge IT investment that has to be embedded into the hospital data center.
 Using healthcare RFID technology, readers track the individual tags on each item and report on its status. Smart cabinets identify the disposition of items every 18 minutes. With multiple readers and redundancy of reads, a continuous read cycle captures the disposition as products are removed or returned to the shelf. No more manual counting.
Using healthcare RFID technology, readers track the individual tags on each item and report on its status. Smart cabinets identify the disposition of items every 18 minutes. With multiple readers and redundancy of reads, a continuous read cycle captures the disposition as products are removed or returned to the shelf. No more manual counting.
Hospital staffs simply wave the item at the Point of Service reader to capture its usage, resulting in more accurate target inventory, product replenishment, and charge capture. Users logon to the internet and view a host of user-friendly reports and analytical tools to effectively manage inventory supply levels. A customer-driven company, WaveMark focuses a lot of energy on customizing the reports to the individual needs of hospitals, so there are generally three or four new web-based software releases annually. But they are all included in the subscription. No more expensive upgrades.
And yes, it is a subscription service, not a capital acquisition. WaveMark retains ownership of the equipment. Hospitals and surgi-centers subscribe to the service, which WaveMark updates continuously. No more technology obsolescence.
Medical OEMs, with lots of product in the field on consignment, are also benefiting from WaveMark CIMS. Historically, these manufacturers have struggled with a lack of visibility to these expensive resources with no way to track expired product. With WaveMark’s solution, they can provide better service to their customers, a more cost-effective supply chain for their shareholders and, ultimately, improved patient safety.
Mack’s involvement
WaveMark has chosen MackMedical as its manufacturing arm. There are currently seven RFID-enabled cabinets in WaveMark’s portfolio, including a five-shelf cabinet, five-shelf cabinet with locking doors, hanging cabinet with six hangers, bin for tall boxes, mobile four-shelf cabinet, tabletop one-shelf cabinet, and a hanging cabinet with doors that is still in development.
All models feature RFID technology and are built of high grade steel and engineering thermoplastics. They are each subjected to a rigorous cycle of quality testing that includes:
 54-step build check sheet to verify proper configuration of critical element
54-step build check sheet to verify proper configuration of critical element- Hi-Pot test to ensure power supplies and safety grounds are correctly configured
- Several software checks for proper configuration and setup
- Physical checks on motors
- Communication/connectivity checks between boards, computers, motors
- Full performance test with calibrated test set to simulate actual use
- 48-hour burn-in with second full performance test
- Verification of communications within cabinet and from cabinet to server
- Final quality audit prior to packaging.
Additionally, all work instructions are formatted with quality checks at key points of subassembly.
Wringing out waste
“The beauty of WaveMark CIMS is that it’s driving waste out of the system that burdens both hospitals and manufacturers,” adds Endicott. Through RFID technology that is turbo-charged by the web, WaveMark is enabling razor-thin inventories and real-time inventory analysis, as well as saving countless hours of time.
On March 23, 2010, The Affordable Care Act was signed into law. “If anything, it will be positive for WaveMark because there’s going to be even more emphasis on the cost-effectiveness of care,” says Endicott. “To the extent that WaveMark has helped our customers reduce cost, increase revenue and improve margins, I think the new health care bill is going to serve us well.”
For more information, contact salesnorth@mack.com.
……………………………………………………………………..
Defribillator Handle
CAE teams with gas-assist for defibrillator solution
The 911 caller complains of chest pain, shortness of breath. EMTs speed to the scene, ambulance defibrillator in tow. When they arrive, assessing and treating the patient is Priority #1. Time is precious and the equipment must perform.
Designed to meet the specific demands and extreme conditions of the emergency medical services environment, this ambulance defibrillator features a suitcase-style design with an innovative protective roll cage, allowing emergency personnel to carry and store the device easily. Lightweight, yet indestructible, it can be dropped off a building or run over by a truck without affecting operation.
 The unit is protected on three sides with gas-assisted parts – two side rails and the front handle, the subject of this case study. The handle is used both to carry the unit and set it up at an angle. Therefore, it has to be durable and sturdy enough to carry the 15-lb device, yet comfortable and easy to grip for the emergency medical technician handling the product. (Image on left: A cross-section of the finished over-molded handle clearly illustrates the gas channel, polycarbonate substrate and TPU overmold.
The unit is protected on three sides with gas-assisted parts – two side rails and the front handle, the subject of this case study. The handle is used both to carry the unit and set it up at an angle. Therefore, it has to be durable and sturdy enough to carry the 15-lb device, yet comfortable and easy to grip for the emergency medical technician handling the product. (Image on left: A cross-section of the finished over-molded handle clearly illustrates the gas channel, polycarbonate substrate and TPU overmold.
The Challenge
A key element of the product’s exterior design is the light, hollow injection molded handle. The customer wanted a handle that was hollow, but stiff enough to pass rigorous tests. Specifications also included a soft-touch feel for easier transport and a snug grip. There could be no gas pinhole in the handle, and both the substrate and overmolding resins had to be capable of developing a chemical bond.
 While hollow, the substrate also had to be structural enough to withstand a shutoff in the steel tool to prevent flash. To accomplish this, the tooled steel had to apply just the right amount of pressure to the substrate to stop the flow of the overmolding resin without crushing the substrate. (Figure on right: The shutoff that defines the boundary between the overmold (black) and substrate results in a grooved design.)
While hollow, the substrate also had to be structural enough to withstand a shutoff in the steel tool to prevent flash. To accomplish this, the tooled steel had to apply just the right amount of pressure to the substrate to stop the flow of the overmolding resin without crushing the substrate. (Figure on right: The shutoff that defines the boundary between the overmold (black) and substrate results in a grooved design.)
“Remember, as you inject the overmolding resin into the tool, the substrate heats up as well,” says Dr. Michael Hansen, senior technical development engineer. “So the substrate must be rigid enough to survive the temperature rise without collapsing. This requires a consistent wall thickness distribution, both for the length of the part and its cross-section.”
CAE Confirms Solution
To produce a handle that will pass all the requirements, the first step is to choose the right gas-assist process.
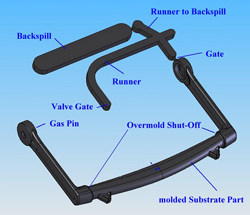 In this case, the best technique is full-shot molding with backspill, which results in a complete filling of the cavity with melt, followed by gas injection and opening of the backspill cavities. (Figure on left: Molding concept for gas-assist handle for automated external defibrillator (AED).
In this case, the best technique is full-shot molding with backspill, which results in a complete filling of the cavity with melt, followed by gas injection and opening of the backspill cavities. (Figure on left: Molding concept for gas-assist handle for automated external defibrillator (AED).
“We used CAE and various flow simulation software to determine the size of the backspill cavity, discreetly position the gas pin, and avoid a hole at the gating location,” says Hansen. “Flow simulations also clearly illustrated the remaining wall thickness distribution of the substrate part after we introduced gas into the process.” (Figure on right: Flow simulation traced the path of the gas channel and illustrated wall thickness distribution.)
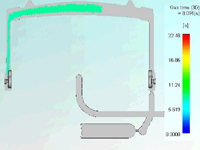 In addition, CAE helped to confirm the processing sequence and cycle time, minimizing material waste and trial and error at the press. CAE also helped to determine a tooling concept with backup options. “For example, machining rather than welding a larger backspill area saves steel, which is costly,” adds Hansen. “In the end, this allows for less expensive tool modifications.”
In addition, CAE helped to confirm the processing sequence and cycle time, minimizing material waste and trial and error at the press. CAE also helped to determine a tooling concept with backup options. “For example, machining rather than welding a larger backspill area saves steel, which is costly,” adds Hansen. “In the end, this allows for less expensive tool modifications.”
The handle is molded in a 300-ton press because of tool size, which incorporates the backspill cavity, valve gate, gas pin, four tool actions and a hydraulic shutoff between the runner and backspill cavity. The substrate is molded of modified polycarbonate resin for chemical resistance, flame retardancy and ECO compliance. It is overmolded with thermoplastic polyurethane for abrasion, required shore hardness, impact resistance, flow length and a comfortable grip.
Mack molds a total of 14 parts for this ambulance defibrillator unit, using thermoplastic polyurethane, polycarbonate and modified ABS resins. All materials are high impact resins with high temperature resistance to assure performance in the harshest of conditions.
For more information, contact salesnorth@mack.com.